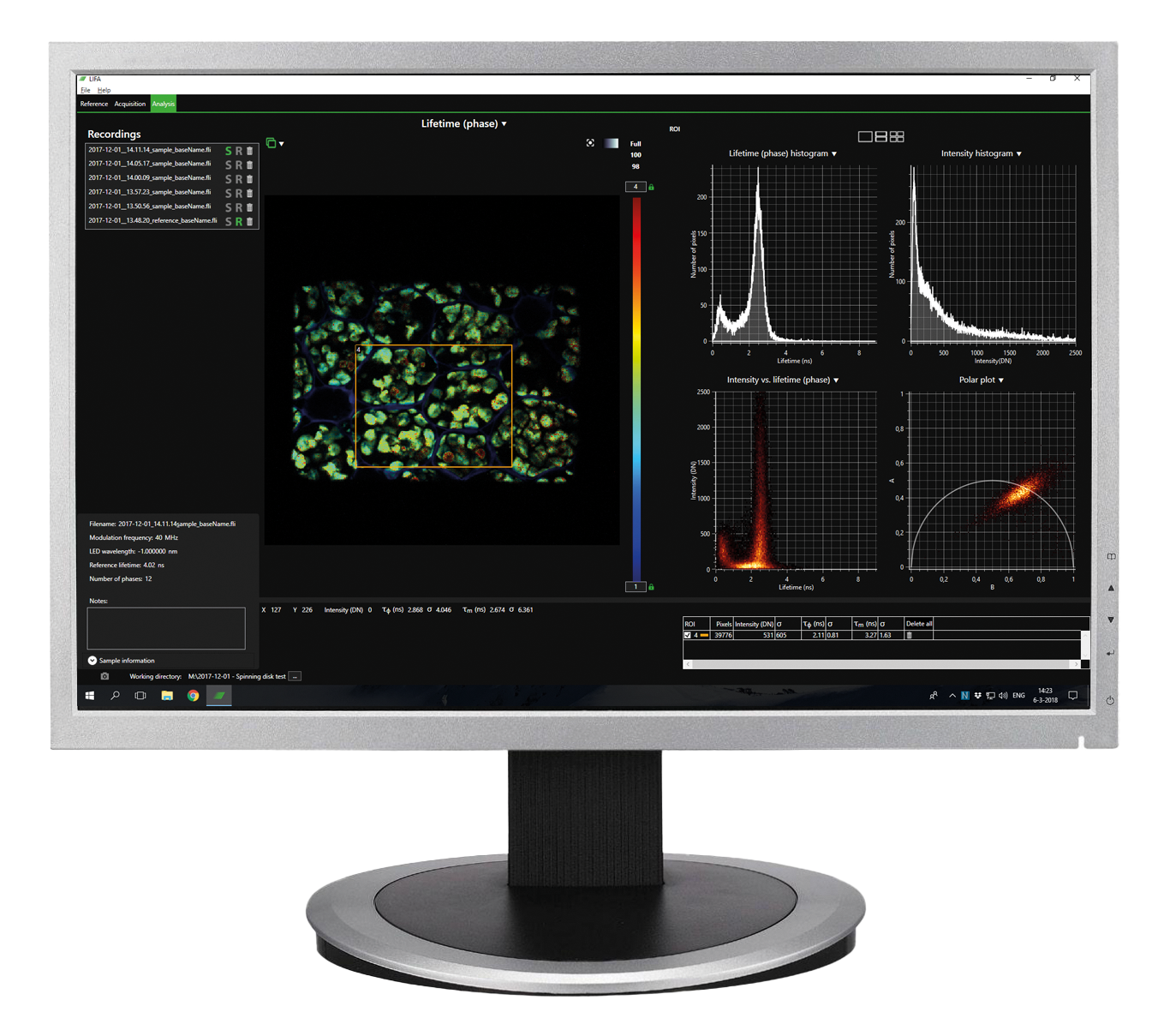
The primary difference between frequency-domain and time-domain FLIM lies in how the fluorescence lifetime is measured:
Frequency-domain FLIM: Modulates the excitation light at specific frequencies and measures the phase shift and modulation of the emitted light relative to the excitation. It determines lifetime from these shifts.
Time-domain FLIM: Uses short light pulses to excite fluorophores and measures the time delay between the excitation pulse and the emitted photons. The fluorescence decay curve is analyzed to determine lifetime.
Both techniques provide insights into molecular environments but differ in how they capture fluorescence data.